Hey everyone! Underneath this post is all of the work that I've done in Section 1 of Chemistry class. We got up to some really cool stuff, so check it out!
|
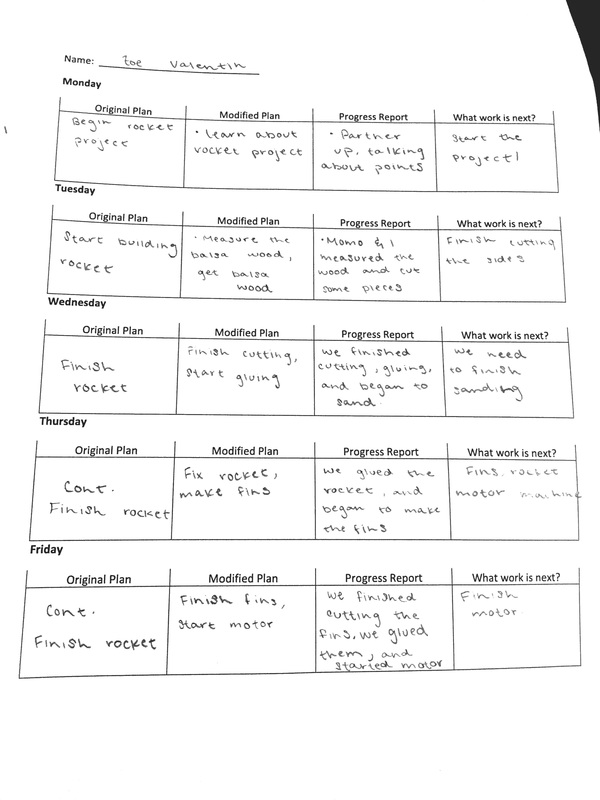
Like my previous 2 week plan, this one describes my plan for each day, the modified plan, progress report, and what needs to be done. This 2 week plan was a little bit easier to fill out because we had a four day weekend. That being said, you will see a lot of modifications throughout the week regarding what we planned and what we actually did. I would say that these past two weeks were really fulfilling. We ended up getting a lot done.
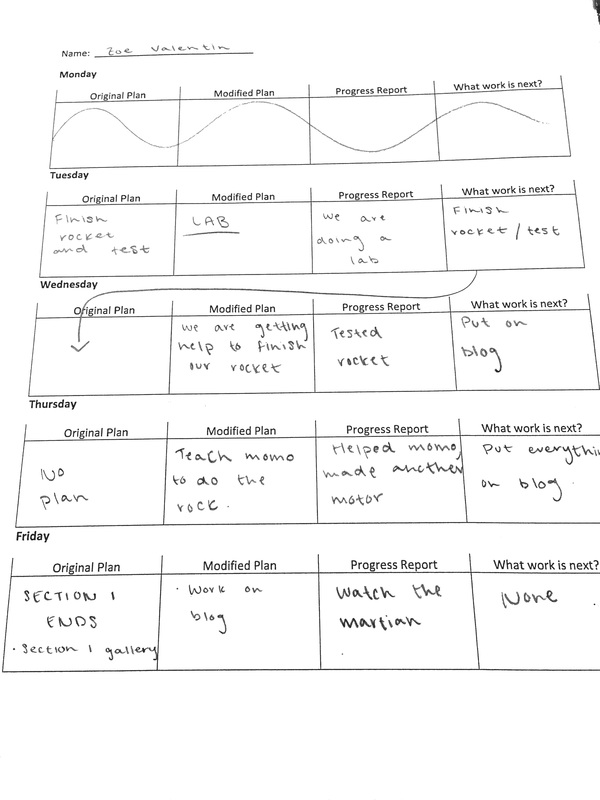
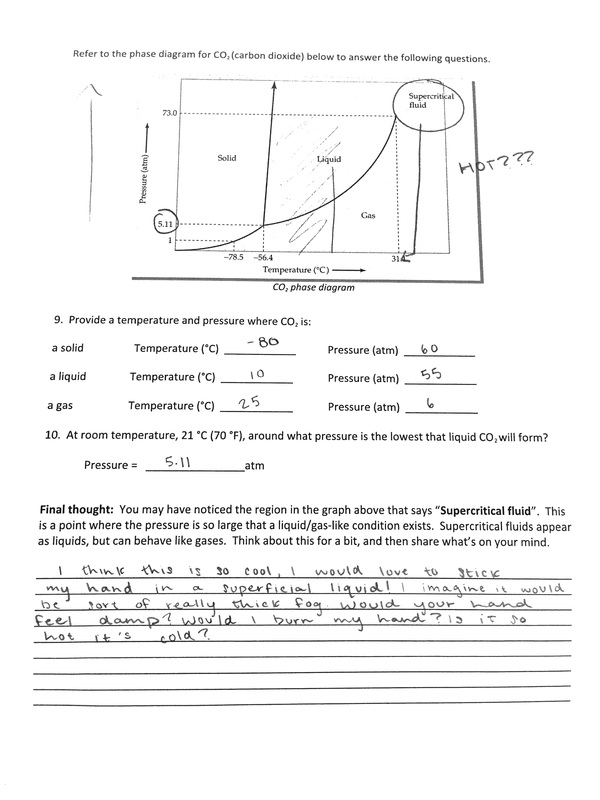
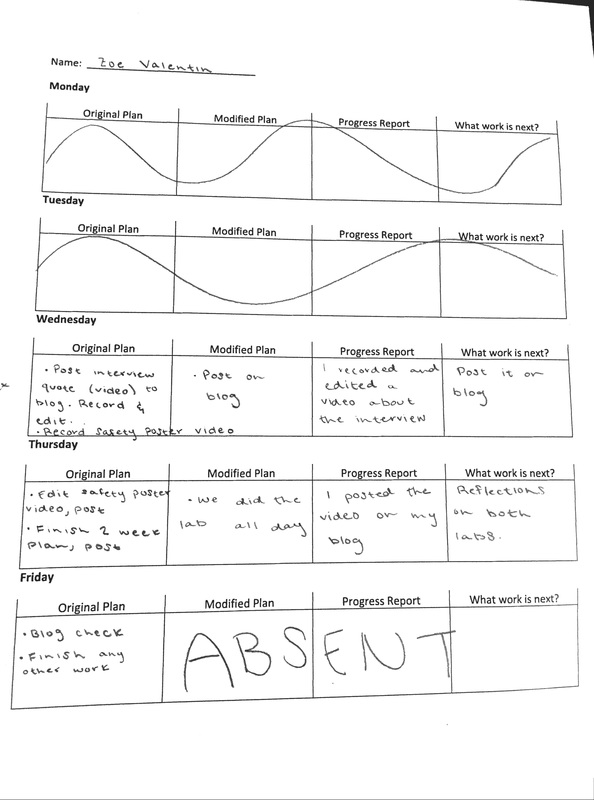
This week, Monique and I partnered up to create our very own...balsa rocket! At first, we found this task a bit daunting. I knew from my experiences last year that rockets weren't my area of expertise (or interest). I think acknowledging this very honest perspective helped me throughout this process. I understood that this was a weakness for me, and I would struggle greatly with building the rocket and understanding its functions as well. Working with Monique has been going smoothly, and I genuinely believe that we push each other to put out our best work. Creating the rocket itself was a bit of a struggle, but we watched our teachers video on how to do it, and it helped us out a lot. Down below I will list out what we accomplished each day. ~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~ 2.2.16 (Tuesday) - Today we began to work on our balsa rocket. We started by gathering the materials we needed. We referred to Andrew's video on how to create them, and decided as a group to follow the measurements he used. We took the balsa wood, and began to measure the sides and trace how we wanted them to look. Andrew showed us how to cut correctly, and we used his advice to cut out the first two sides. We quickly learned that you need a straight edge to help you cut, because some of our sides didn't come out as nicely as we hoped. Our teacher helped us out again, and we re-cut and edited our design. 2.3.16 (Wednesday) - Today Monique and I continued to cut out the sides to our rocket. Since we had Andrews help, it went a lot smoother. We weren't really happy with the sides of the rocket, but we learned that we would sand it down anyways. The next step was super gluing. We re-watched the instructional video, and got help from our teacher again. The first side we glued, we ended up gluing at an angle. Admittedly, it was my fault, and in turn it made out whole rocket look lopsided and weird. I quickly realized the mistake, and we were able to salvage the rocket by taking the sides apart again and gluing them back on in a normal square shape. It was difficult to figure out which piece we had to glue on top or bottom, but we managed to get it done. By the end of third period, we were able to begin sanding it down to create a smooth rocket shape. 2.4.16 (Thursday) - Monique and I continued to work on sanding our rocket down. We quickly learned that if you sand it too much on the wrong spot, it creates cracks in the design. We had to take some sawdust and super glue it into the cracks so the rocket would be able to function properly. I would say that today was the most difficult day by far, because our rocket kept coming apart. I felt bad because at one point, checking to make sure that the rocket was stable, I cracked the entire thing. Three of the sides came apart. This made me feel horrible, because we were just celebrating the fact that we had finally finished. In order to fix this, we took it back inside and got our teachers help to put it back together. Monique was a little worried, and put some super glue all down the inside of our rocket. This caused it to drip from the tip, so we had to wait a little longer for it to dry. While we were waiting, we decided to design and cut out the design of our fins. With a little bit of Avery's help, we were able to cut out two pretty awesome looking fins. 2.5.16 (Friday) - Today Monique and I picked up where we left off, and cut out the other two fins for our rocket. For some reason we were having a lot of trouble with it, and we messed up a lot. Luckily we managed to cut out two more identical fins. When we completed this, we super glued them onto the body of our rocket. Badaboom! We were finished! We then got two small pieces of wooden blocks, and began to make our mini motor machines. We got some help from our peers, and Monique drilled two holes into the side for one part of the hinge. We asked for advice from our teacher, but he suggested we move the holes up a little. He ended up helping us out by drilling the holes. He said that it would work even better than some of our peers, because it would put more pressure on the motor itself when we had to pack it. After the holes were drilled, we had to wait awhile for the screwdriver that was being passed around. Luckily, by the end of the period we were able to screw in one side of the hinge. 2.8.16 (Monday) - In class, we broke our rocket in the first ten minutes. Monique had knocked a fin loose when she had put her backpack in her cubby, and I accidentally hit another fin off when I was talking to our table group. This made us have to reglue the rocket. After this, we worked a bit on our mini rocket motor machine. We were able to screw in the other side of the hinge. Our teacher said that he would build us a latch for it, so we had to wait in line for a long time. After this, he said our machine was a bit off because there was a gap. Tomorrow we will find out if we have to redo the hinge in order to fix it. 2.9.16 (Tuesday) - Today we did a combustion lab, so unfortunately we made no progress on our rocket. 2.10.16 (Wednesday) - Today we spent both periods watching the movie "The Martian". It was awesome. Still, no progress was made on our rocket. 2.11.16 (Thursday) - Today, we did an extension of the combustion lab. We made our very first rocket motor! We were very happy to learn about the main components of it, the fuel, the tools needed to make it, how to use those said tools, and how to put it all together. Monique and I struggled greatly trying to make the motor, and constantly had to get help from our teacher, who was luckily only a few feet away. To our great surprise, when we went out to test it, it was one of the motors that flew the highest. At first we were ecstatic: We had did it! Then we felt a little bummed out because we didn't think to stick the motor in our actual rocket. By the end of third period, we were able to recreate the motor we had just made, but we realized that we were missing a piece of our rocket. We hadn't glued the little piece of wood that is supposed to be inside our rocket to hold it in place when it is launched. Our goal is the get that done by Tuesday, and launch our rocket by then as well. 2.16.16 (Tuesday) - Today we spent both periods doing another lab. 2.17.15 (Wednesday) - Today during first period, Monique and I were able to finally finish our rocket. We put in the little piece of wood to hold the motor in place, and our teacher helped us put on the little staples. We launched it, but it didn't do well. We tried again during third period. FINAL REFLECTION: Today we got to finally test out our rocket! This was definitely a long time coming, and we were relieved and excited to finally get it done. We had struggled getting all of the pieces together and completing the project, so it was nice to see it come to an end. During first period, we were able to launch. When our teacher ignited the fuse, it immediately started to smoke. It shot up a couple of feet and landed on the ground. Our teacher had said that this was because the cap of the motor blew off and the motor itself was old. We had built it last week, so it was sitting out collecting dust for almost 6 days. When we learned of this, Monique and I decided we would build another motor and try again in third period. We did this, and I was surprised to see how easy it all was. I discovered that I really liked making the rocket motors, and I enjoyed helping out my tablemates. When Monique and I were to test our rocket again, we were really nervous. At that point, we just wanted to get a good result so we could leave this mini project feeling satisfied with all of our hard work. This time, the rocket flew up into the air. It was so awesome to see, because all of the other groups didn't do so well and we thought ours wasn't going to as well. Our rocket hit the ground pretty hard, but we were surprised to see that it was perfectly fine. I think that building this rocket was an amazing experience. Monique and I really didn't think we would be interested in the whole rocket part of this section, but I think I can safely say that it wasn't so bad after all. We learned a lot about the craftsmanship behind it, as well as the science. The (rocket) science. ;) I came a little late to class today, so I was surprised to see that we were doing an unplanned lab to start off the period. I wasn't really sure as to what was going on, but I could see that my groupmates had this weird goo consistency in a cup on our table. I played with it for awhile, and was curious to see what was in store for this class. I wasn't sure why, but I knew that I saw what I was playing with before. I just couldn't recall why and where. At this notion of thought, I was intrigued. What was this? I soon learned that before I had walked in, my group had poured water over a couple of grams of sodium polyacrylate. I wasn't sure what that was, but I was interested in learning. We went over the basic science of it ( it is a cross linked polymer of organic salt) before we jumped into the lab. I was able to break down the word, which helped me in understanding the molecule better. A polymer is a bunch of monomers linked together, a long chain of repeating units. Cross linked is something intertwined with itself. Then we went over different bonds, which would help us absorb the lab better (did you catch the pun?). Dashes are covalent bonds (share a pair of electrons with each other). It's a fairly weak bond. Since they are so weak they tend to be a gas or a liquid but if there's enough of it, it becomes a solid. A polar covalent bond is a neutrally charged molecule. It causes the oxygen to be slightly negative and the hydrogen slightly positive. Ionic bonds are very strong and very polar. It causes the oxygen to be very negatively charged and the sodium to be very positive. Once this was established, we were ready to start our lab. Looking Back: What happened? For this lab, we were to find out how much water the sodium polyacrylate can absorb before it turns into a liquid again. I would say that my group had its ups and downs with it, but we found a reasonable conclusion. I think our experiment turned out pretty cool, and there were different stages that we went through to answer the essential question. We tried many different things throughout our lab, like adding Gatorade to our substance, food coloring, etc. We added a total of 600 ml to the 4 grams of sodium polyacrylate. Overall, I would say that the lab was pretty successful in answering the question, but it was the most successful in terms of the knowledge gained. Looking Beyond: What do you think has happened? What’s the science? I think I was the most excited to report back on my blog for this specific question. I never truly understood what was going on in any of the other labs besides this one. I honestly feel like I know this information in my head, and understand it on so many levels. I came to the conclusion that due to osmosis, a solvent tends to move from areas of low solute concentration to areas of high solute concentration. In our case, the solvent is water and the solute is sodium. The water wants to move inside to even out the concentration. The molecule is expanding because the water is being forced in, which is why we get the gel consistency. Sometimes this is called water lock, since it can absorb so much. At the end of the lab, we were able to pour salt on our solutions to turn them back into a liquid. When you add the salt, the high sodium in concentration is outside, so the water wants to rush out again. I thought this was pretty cool. Looking inward: Humans doing science I would say that for the most part, we were able to get the desirable results. There were many things I wished we could do differently throughout this lab, but that was mostly because of the group I worked with. I think that we were able to answer the essential question efficiently, so that's good. I think I performed really well in this activity because I helped out my group a lot. I made sure that we were always aiming to answer the question, and get back on track. Looking outward: Collaboration and Impact I worked with my normal table group for this lab. I would say that it was pretty difficult this time around, which sucks because I was just beginning to change my view on the group dynamics. It was hard to get everyone in my group to focus on the task at hand, which was figuring out how much water the sodium polyacrylate could take. For a couple members of my group it seemed like play time. They just wanted to mess around with it a lot. I get that the experimentation part is supposed to be fun, but there's still a goal that is trying to be met. It caused a lot of frustration within the group, and although not spoken aloud, it was evident of the tension. For my group, I made sure to take lots of videos and help out as much as possible. Looking Forward: What next? If I could do this lab over again, I would probably change some of the people I worked with. I know who works hard in my group, as well as who doesn't. It was very evident today. I think I would enjoy doing this lab again just to do it. It was very fun, and making the connections between what we learned and what was taking place during the lab was awesome. This really made me want to figure out what else I can do with the awesome solution. Take Aways: An extra tidbit I learned was that it can absorb 400 times its mass, mineral content makes a big difference,etc. I also wondered: What happens when you add pressure to the concoction? Does the water escape? Today, we had some downtime in building our rocket. We waited for quite a while to get our latch, so I decided to do a worksheet. Luckily, the worksheet that I found was on a topic that we had covered last week. Still, it was a bit challenging. I had to look up some of the key words and what they meant, so I could rightfully answer the questions. One thing that stuck out to me while doing these problems was the last question. It was referring to the phase diagram for carbon dioxide, and was asking about superficial liquid. That is when the pressure is so large that a liquid/gas-like condition exists. I was really impressed thinking about this, but madly curious at the same time. I couldn't really wrap my head around it. Also, according to the diagram, it would be immensely hot. I thought it would be like a thick fog, and amusedly wrote down that I "wanted to stick my hand in it". Seems a bit silly now. Anyways, the worksheet is down below.
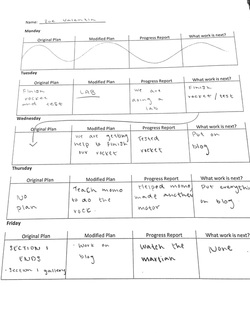
Today in class, I was excited to learn the day's lesson. I was always interested in how the Periodic Table worked, and how it all came together. Luckily for me, we were able to learn about just that. We started off by breaking it all down. Quite literally. Right to the atom. Atoms are the smallest unit of an element, so it was really cool to uncover the structure of one, and how it played a part in combining elements. That wasn't the only structure we learned about. We were informed on how the Periodic Table is broken up, which is by the number of energy levels an atom of an element has and the number of electrons in the outer energy level. Our teacher gave us some interesting examples, and we were able to figure out how the structure of sulfur would look, and how to make a nitrogen with two magnesium's. After we absorbed the information a little more, we were able to start our lab: Combustion Reaction. Looking Back: What happened? During our experiment, we were able to combine citric acid and water to a flask. We added strips of magnesium to it, and quickly covered it with a balloon. Once we did this, we were surprised to see that the balloon was beginning to expand. A little after this, the bottom of the flask began to heat up a bit. We decided to put it in a bowl of water to cool it down. When we thought our balloon was a decent size, we took it off of the flask and tied it off. Immediately after this, my group and I noticed that the balloon had the ability to float. This shocked us, and we found the lab much more interesting. How was this able to happen? Why wasn't it weighted down like oxygen? Once everyone in the class had their balloons tied off, we turned off the lights and tested them out. We pinned our balloons to a ruler (one at a time) and stood back. We hovered them over a candle until they popped. The reaction was immediate. It exploded in a ball of fire. In the end, we were able to make a little bit of water. Looking Beyond: What do you think has happened? What’s the science? After this experiment, I was a little curious. I knew that in our lab, atoms were combining to create new substances with new properties. I found this interesting. I knew that by putting the little strips of magnesium into the citric acid and water, chemicals were reacting. This explained why the balloon ended up inflating, and the reaction it had to the heat. We had created hydrogen gas. We learned that by adding the citric acid and water, we were creating hydrogen, and by adding the magnesium it was giving the hydrogen extra electrons, which was making the gas. I found this really cool, and it made me wonder at the end of the lab: What is fire then? What is that reaction? Our teacher explained that when atoms come into energy, it absorbs it and the electrons jump. When there is no more energy, the electrons go back down and release the energy (light and heat). I later found out that the energy is released at specific times. This is why the flame is above the reaction point. Looking inward: Humans doing science I would say that my group did a great job with this lab. We ended up getting all of the results we wanted. We successfully inflated the balloon with hydrogen gas, unlike some other groups who ended up getting water in their balloons. We also were pleased with how well the balloon reacted with the fire. It did everything it was supposed to. I would say that this was the smoothest lab that I have been able to complete in this class. Nothing happened that was not planned in advance. Looking outward: Collaboration and Impact In this lab, I was able to work with Monique, Hannah, Rebekah, and Fatuma. I would say that today we all worked really well together. No one goofed off too much, and we were able to pull off a really awesome experiment. Today my group performed really well, and it made me revisit the idea of working with new people. I can see the strengths and weakness' of working with the people I did, and I can acknowledge them rightfully. I contributed to the group by taking lots of videos, and editing them all together. I would say that making the videos for my group is something that I like to do for them. I know that it helps with everyone's blogs a lot, and even though its a lot of extra work, I'm glad to help them out. Looking Forward: What next? If I could do this again, I would want to wait a little while longer for our balloon to fill up. It was a bit on the smaller size, so the reaction and fire ball wasn't that big. If I were able to work with anyone to do this lab again, I would probably want to work with just Monique. I feel like we work well together, and sometimes that can get washed out when we have to work with a larger group. We both ride the same wavelength, so when one of us is getting annoyed with someone in our group, odds are the other is feeling the exact same way. Since we are so in tune, I feel like these labs would be beneficial just to have the two of us do them. Take Aways: Doing this lab was definitely really fun. I had a connection to it, because my teacher made a hydrogen balloon for my birthday, and had me pop it in front of the whole class using the candle. I thought it was super cool, and I was intrigued on how it worked. I'm glad I was able to see on a molecular level, the reactions taking place. Also, two kids who were in my class last year did this same lab, and I always wondered how they even came across something like making hydrogen balloons. It was almost unheard of for me. Now I can see that it's not so crazy after all, and instead is super cool. As always, down below is the video I made on this lab. Enjoy! Hey everyone! In class, we write up and complete 2 week plans, which help us stay organized. Down below are documents of my very first plan, which was from January 27th until February 5th. Each day has an original plan (what we planned to do in advance) a modified plan (what seems more realistic to that day) a progress report (what we are actually doing that day) and a what work is next part (self-explanatory).
In class on Thursday, we experimented with dry ice. We were trying to find out how to calculate the volume of a gas from a solid.
Looking Back: What happened? Looking back, I would say that our experiment went pretty well. We tried to calculate the volume of a small piece of dry ice, and to do it we put it in a balloon. We tied it off, and by the time it melted it created a small balloon. My group also messed around a lot, and tried different things with the dry ice. Looking Beyond: What do you think has happened? What’s the science? Whilst experimenting, I was shocked to see how the balloon was expanding. I thought maybe the ice would freeze the balloon, but that didn't happen. I was confused on how the next steps we needed to take once the balloon expanded, in order to measure the volume. I understood that there was an equation, but we had failed to measure the piece of dry ice before hand. This made it difficult to make connections between the volume of a gas and a solid and the difference between the two. Looking inward: Humans doing science If I could do this experiment again, I would want to make sure we used a bigger piece of dry ice. The piece of ice we used was pretty small, so it didn't create the results we wanted. I would also want to make sure that we measure the piece before hand, so we can actually complete the experiment. Looking outward: Collaboration and Impact The group that I worked with was one that I worked with before. It was Monique, Sophie, Rebekah, and Fatuma. It was difficult because some of the members of my group weren't on task, and didn't know the point of the experiment. They were more interested in playing with the dry ice, and their own endeavors with it. This made me a little frustrated, because the point of the lab really interested me, and I genuinely wanted to understand the essential question. I think that if I had to work on a lab or project in Chemistry moving forward, I would want to change some of the people I worked with. Looking Forward: What next? Overall, I really enjoyed this lab and I learned a lot. It intrigued me how dry ice worked, and it was interesting to work with to say the least. I liked how many different things we were able to do with this lab, and all of the directions we could have taken it to. Down below are some pictures from the lab. Enjoy! Last Wednesday, our class did a lab where we got to create Oobleck. At first, I had no idea what that was, but upon explanation, I realized that I have made Oobleck loads of times without knowing it. This got me excited to get to work. Looking Back: What happened? Looking back, I would say that our early trials of creating Oobleck wasn't as expected. We understood the ratio (3:1 - Oobleck to water) but we still felt as though it was too dry. We were very stingy with the water at first, so it made to substance powdery and hard. Then we tried pouring more in, which created a puddle. But by the end, our Oobleck was perfect. Looking Beyond: What do you think has happened? What’s the science? While we were experimenting, I was very interested in how the substance was acting. When I put some on my hands, it almost seemed like a solid. Seconds later, it would melt and drip between my fingers like it was a liquid. This seemed weird to me, and I wondered how it changed its state so quickly, and what was happening to get that reaction. Looking inward: Humans doing science Everyone in my group was successful in making their own stressball with our Oobleck. This was because by the time we were finishing up, we had made our Oobleck the right texture and substance. Like I said before, we struggled at the beginning, but we managed to figure it out through trial and error. This gave us desirable results. Looking outward: Collaboration and Impact For this lab, I worked with Monique, Hannah, and Sophie. This was perfect, because we worked together as a group for our last project, and it turned out really great. We understood how everyone in the group worked, and we were not afraid to speak our minds and probe our curiosity. Throughout the lab, I contributed by taking videos of our process, which helped out my group in documenting what we were doing. Looking Forward: What next? If I could do this lab again, I would want to spend more time studying the Oobleck. I think I was too worried about whether we were doing it right to really delve into what was going on. I very much liked the people who I worked with, and I think that we make a great team for these kinds of things. Overall, I think the lab was very fun and it taught us to be curious about the forms of matter and how they worked. |
Zoe Valentin"I'm gonna have to science the shit out of this." -Mark Watney (The Martian) Archives
June 2016
Categories |







 RSS Feed
RSS Feed
